- Nevirapine
-

- $1.00 / 1ASSAYS
-
2025-03-28
- CAS:129618-40-2
- Min. Order: 1ASSAYS
- Purity: 99
- Supply Ability: 500
- Nevirapine
-

- $0.00 / 1KG
-
2025-03-16
- CAS:129618-40-2
- Min. Order: 100g
- Purity: 98%+
- Supply Ability: 100kg
- Nevirapine
-

- $1.00 / 100G
-
2025-01-08
- CAS:129618-40-2
- Min. Order: 100G
- Purity: 99%
- Supply Ability: 50000tons
|
| | Nevirapine Basic information |
| Product Name: | Nevirapine | | Synonyms: | 6H-DIPYRIDO[3,2-B:2',3'-E][1,4]DIAZEPIN-6-ONE, 11-CYCLOPROPYL-5,11-DIHYDRO-4-METHYL-;11-Cyclopropyl-4-Methyl-5H-dipyrido[3,2-b:2',3'-e][1,4]diazepin-6(11H)-one;6H-Dipyrido[2,3-b:3',2'-e][1,4]diazepin-6-one, 11-cyclopropyl-5,11-dihydro-4-Methyl-;AIDS005653;BI-RG-587 & CD4-IgG;D00435;MLS00008458;N11-Cyclopropyl-4-methyl-5,11-dihydro-6H-dipyrido[3,2-b:2',3'-e]-[1,4]diazepin-6-one & CD4-immunoadhesin | | CAS: | 129618-40-2 | | MF: | C15H14N4O | | MW: | 266.3 | | EINECS: | 603-345-0 | | Product Categories: | Other APIs;API;Anti-virals;Inhibitors;Intermediates & Fine Chemicals;Non-nucleoside Reverse Transcriptase;Pharmaceuticals;Active Pharmaceutical Ingredients | | Mol File: | 129618-40-2.mol | 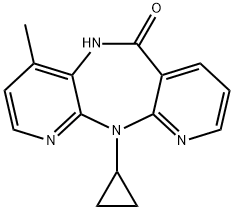 |
| | Nevirapine Chemical Properties |
| Melting point | 247°C | | Boiling point | 409.5°C (rough estimate) | | density | 1.1300 (rough estimate) | | refractive index | 1.6200 (estimate) | | Fp | 9℃ | | storage temp. | 2-8°C | | solubility | DMSO: ≥22mg/mL | | form | powder | | pka | 2.8(at 25℃) | | color | white to tan | | Water Solubility | 0.1g/L(temperature not stated) | | Merck | 14,6490 | | BCS Class | 2 | | CAS DataBase Reference | 129618-40-2(CAS DataBase Reference) | | EPA Substance Registry System | |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-36-37/39 | | RIDADR | UN1230 - class 3 - PG 2 - Methanol, solution | | WGK Germany | 2 | | RTECS | JM5562500 | | HS Code | 29339900 | | Hazardous Substances Data | |
| | Nevirapine Usage And Synthesis |
| Description | Nevirapine and its analogues exhibit antiretroviral effect against azothymidine-resistant HIV strains.
Nevirapine in combination with ZDV and ddI produced approximately 18% higher CD4 cell counts and a
decrease in viral load compared with patients who took ZDV and ddI. Nevirapine is recommended with
nucleosides for patients infected with HIV-1 who have experienced clinical or immunologic deterioration.
The significant side effects of nevirapine are liver dysfunction and skin rashes. | | Chemical Properties | Crystalline Solid | | Originator | Neve,Le Sante,India | | Uses | amyloidosis therapy | | Uses | A potent (IC50=84nM) and selective non-nucleoside inhibitorf HIV-1 reverse transcriptase | | Uses | Labelled Nevirapine , a potent (IC50=84nM) and selective non-nucleoside inhibitor of HIV-1 reverse transcriptase. Antiviral.;Labeled Nevirapine, intended for use as an internal standard for the quantification of Nevirapine by GC- or LC-mass spectrometry. | | Definition | ChEBI: A dipyridodiazepine that is 5,11-dihydro-6H-dipyrido[3,2-b:2',3'-e][1,4]diazepine which is substituted by methyl, oxo, and cyclopropyl groups at positions 4, 6, and 11, respectively. A non-nucleoside reverse tr
nscriptase inhibitor with activity against HIV-1, it is used in combination with other antiretrovirals for the treatment of HIV infection. | | Indications | Nevirapine (Viramune) is approved for the treatment of
HIV infection in adults and children as part of a combination
therapy. During the first 12 weeks of treatment,
patients must be closely monitored for the development
of potentially fatal hepatic toxicity (i.e., hepatitis, hepatic
necrosis, and hepatic failure) and skin reactions (i.e.,
Stevens-Johnson syndrome, toxic epidermal necrolysis,
and hypersensitivity reactions). Although these toxicities
are rare, common side effects include mild to moderate
rash, fever, nausea, fatigue, headache, and elevated
liver enzymes. | | Manufacturing Process | There are 3 ways for preparing of nevirapine.
117.5 kg of 2-chloro-N-(2-chloro-4-methyl-3-pyridyl)-3-pyridine carboxamide,
23.3 kg of calcium oxide and 59.4 kg of cyclopropylamine (molar ratio:
1:1:2.5) are heated to between 135° and 145°C in 235 L of diglyme
(diethylene glycoldimethylether) in a 500 L VA autoclave over a period of 6 to
8 hours. The reaction mixture is then cooled to a temperature of 20°-30°C
and filtered. The filter cake is washed with 58.8 L of diglyme. The filtrates are
combined and initially 200 L of solvent is distilled off. The residue is then
diluted with a further 117.5 L of diglyme. The resultant diluted solution is
added over a period of 20 to 40 minutes to a suspension of 45.0 kg of 60%
sodium hydride in 352.5 L of diglyme, heated to 130°C. The storage vessel
and conduits are rinsed with a further 55.8 L of diglyme, and the mixture is
stirred at a temperature of between 130° and 140°C for a further 30 to 60
minutes. The majority of the diglyme is then distilled off. Finally, the remaining residue is carefully mixed with 470 L of water. After cooling to a
temperature of about 25°C, 235.0 L of cyclohexane and 57.11 of glacial acetic
acid are added to the reaction mixture. The mixture is then stirred for about 1
hour at temperature of 10° to 25°C. The resultant suspension is centrifuged
and the centrifuged material is then washed with 235.0 L of methyl-tert�butylether and subsequently with 353.5 L of water and finally with 235 L of
ethanol. In this way, after drying, 92.5 kg (83.5% of theory) of 11-
cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3,2-b:2',3'-e][1,4]diazepin-6-
one (nevirapine) is isolated.
117.5 kg of 2-chloro-N-(2-chloro-4-methyl-3-pyridyl)-3-pyridine carboxamide,
46.7 kg of calcium oxide and 47.5 kg of cyclopropylamine (molar ratio: 1:2:2)
are heated to 135° to 145°C in 235 L of diglyme (diethylene glycol
dimethylether) in a 500 L VA autoclave over a period of 6 to 8 hours. The
reaction mixture is then cooled to a temperature of 20° to 30°C and filtered.
The filter cake is washed with 58.8 L of diglyme. The filtrates are combined
and about 188 L of solvent is distilled off. The residue is then diluted with a
further 117.5 L of diglyme. Over a period of 20 to 40 minutes, the resultant
diluted solution is added to a suspension of 45.0 kg of 60% sodium hydride in
352.5 L of diglyme, heated to 130°C. The storage vessel and conduits are
rinsed with a further 55.8 L of diglyme and the mixture is stirred at a
temperature of 130° to 140°C for a further 30 to 60 minutes. The majority of
the diglyme is then distilled off. Finally, the remaining residue is carefully
mixed with 470.0 L of water. The reaction mixture is cooled to a temperature
of about 25°C and 235.0 L of cyclohexane and 57.1 L of glacial acetic acid are
added. The mixture is then stirred for about 1 hour at a temperature of 10o to
25°C. The resultant suspension is centrifuged and the centrifuged material is
washed with 235.0 L of methyl tert-butylether, followed by 353.5 L of water
and finally with 235 L of ethanol. In this way, after drying, 90.6 kg (81.7% of
theory) of 11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido-[3,2-b:2',3'-
e][1,4]diazepin-6-one (nevirapine) is isolated.
287.2 kg of 2-chloro-N-(2-chloro-4-methyl-3-pyridyl)-3-pyridine carboxamide,
57.0 kg of calcium oxide and 87.1 kg of cyclopropylamine (molar ratio:
1:1:1.5) are heated in 574 L of diglyme (diethylene glycol-dimethylether) to
135°-145°C for about 30 minutes in a 1200 L VA stirring apparatus. This
produces a pressure of 1.2-1.5 bar and about 50% of the starting material is
reacted. To this mixture, over about 30 minutes at 135°-145°C, a further 58.1
kg of cyclopropylamine is added producing a pressure of 3.0-3.5 bar, and
another 25% of the starting material is reacted. The mixture is then kept at
135°-145°C for a period of 5 to 6 hours. The reaction mixture is then cooled
to a temperature of 20° to 30°C and filtered. The filter cake is washed with
144 L of diglyme. The filtrates are combined and 400 L of solvent is distilled
off. The residue is then diluted with a further 287 L of diglyme. Over 20-40
minutes, the resultant diluted solution is added to a suspension of 110 kg of
60% sodium hydride in 862 L of diglyme, heated to 130°C. The storage vessel
and conduits are rinsed with a further 144 L of diglyme and the mixture is
stirred at a temperature of 130° to 140°C for another 30 to 60 minutes. The
majority of the diglyme is then distilled off. Finally, the remaining residue is
carefully mixed with 1150 L of water. After the reaction mixture has been
cooled to a temperature of about 25°C, 575 L of cyclohexane and 147 L of
glacial acetic acid are added. The mixture is then stirred for about 1 hour at a
temperature of 10°-25°C. The resultant suspension is centrifuged and the
centrifuged material is then washed with 575 L of methyl-tert-butylether,
followed by 862 L of water and finally with 575 L of ethanol. In this way, after drying, 225 kg (83.0% of theory) of 11-cyclopropyl-5,11-dihydro-4-methyl-
6H-dipyrido[3,2-b :2',3'-e][1,4 ]diazepin-6-one (nevirapine) is obtained. | | Brand name | Viramune (Boehringer Ingelheim);Nevimune. | | Therapeutic Function | Antiviral | | Acquired resistance | One or more changes within the HIV reverse transcriptase at
amino acid positions 100, 103, 106, 108, 181, 188 and 190
are associated with resistance. These point mutations have also
been implicated, either alone or in combination, in HIV resistance
to other non-nucleoside reverse transcriptase inhibitors. | | General Description | Nevirapine (Viramune) is more than 90% absorbed by theoral route and is widely distributed throughout the body. Itdistributes well into breast milk and crosses the placenta.Transplacental concentrations are about 50% those ofserum. The drug is extensively transformed by cytochromeP450 (CYP) to inactive hydroxylated metabolites; it mayundergo enterohepatic recycling. | | Pharmaceutical Applications | A synthetic heterocyclic compound formulated for oral use as
anhydrous compound or as the hemihydrate in a liquid oral
suspension. | | Biochem/physiol Actions | Nevirapine is an allosteric, non-nucleoside inhibitor of HIV reverse transcriptase (NNRTI). The Ki for inhibition of wild-type RT by Nevirapine is 200 nM. | | Mechanism of action | Nevirapine is a dipyridodiazepinone derivative that binds directly to RT . Thus, it blocks RNA- and
DNA-dependent polymerase activities by causing a disruption of the enzyme's catalytic site. The activity
of nevirapine does not compete with template or nucleoside triphosphate. The HIV-2 RT and human DNA
polymerases are not inhibited by nevirapine. The 50% inhibitory concentration ranged within 10 to 100
nM against HIV-1. | | Pharmacokinetics | Oral absorption: c. 93%
Cmax 200 mg twice daily: c. 5.74 mg/L
Cmin 200 mg twice daily: c. 2.88 mg/L
Plasma half-life: c. 36 h
Volume of distribution: c. 1.21 L/kg
Plasma protein binding: c. 60%
Absorption and distribution
Nevirapine is orally very well absorbed and widely distributed. CNS penetration is good and the semen:plasma ratio is in the range of 0.6–1. It is distributed into breast milk.
Metabolism and excretion
It is extensively metabolized by cytochrome P450 enzymes into a number of hydroxylated intermediates that are subsequently conjugated with glucuronide. Around 81% of the dose is excreted in urine (<5% as unchanged compound) and 10% in feces. There is no significant change in the pharmacokinetics in renal impairment. It is contraindicated in patients with severe hepatic impairment; caution should be exercised in patients with moderate hepatic dysfunction. | | Clinical Use | Treatment of HIV-1 infection in adults and children over 2 months old (in
combination with other antiretroviral therapies)
Reduction of maternal transmission of HIV to the fetus (recommended
only for use in HIV-infected treatment-naive women in labor who have
had no prior HIV therapy) | | Side effects | Life-threatening hepatic events, including fulminant hepatitis, have been observed in treatment-naive patients, generally within the first few weeks of treatment, but sometimes later. Approximately half the patients also develop skin rash, with or without fever or constitutional symptoms. Women with elevated CD4 counts (>250 cells/mm3) appear to be at highest risk. Men with pretreatment CD4 counts >400 cells/mm3 are also at increased risk. These risks exist in the absence of underlying hepatic abnormalities and, in some cases, hepatic injury continues to progress despite discontinuation of treatment. Treatment should stop, and not be restarted, in patients with clinical evidence of hepatitis. A starting dose of 200 mg per day, with escalation to full dose if no adverse reaction occurs, reduces the frequency of reaction. Single doses given to mothers or infants for prevention of perinatal HIV infection appear safe. | | Drug interactions | Potentially hazardous interactions with other drugs
Antibacterials: reduces concentration of
clarithromycin, but concentration of active
metabolite increased, also concentration of
nevirapine increased; concentration decreased by
rifampicin - avoid; possibly increased rifabutin
concentration.
Anticoagulants: may increase or reduce effect of
warfarin.
Antidepressants: concentration reduced by St John’s
wort - avoid.
Antifungals: concentration of ketoconazole reduced
- avoid; concentration increased by fluconazole;
possibly reduced caspofungin and itraconazole
concentration - may need to increase caspofungin
and itraconazole dose.
Antipsychotics: possibly reduced aripiprazole
concentration - increase aripiprazole dose.
Antivirals: concentration of dolutegravir, indinavir
and efavirenz reduced and possibly etravirine,
fosamprenavir, lopinavir, simeprevir and atazanavir
- avoid with atazanavir, etravirine and simeprevir
consider increasing lopinavir dose; increased risk of
granulocytopenia with zidovudine.
Cytotoxics: avoid with olaparib.
Guanfacine: concentration possibly reduced -
increase guanfacine dose.
Oestrogens and progestogens: accelerated
metabolism (reduced contraceptive effect).
Orlistat: absorption possibly reduced by orlistat.
Ulipristal: possibly reduces contraceptive effect. | | Metabolism | Nevirapine is extensively metabolised by hepatic
microsomal enzymes, mainly by the cytochrome P450
isoenzymes CYP3A4 and CYP2B6, to several inactive
hydroxylated metabolites. Auto-induction of these
enzymes results in a 1.5- to 2-fold increase in apparent
oral clearance after 2-4 weeks at usual dosage, and a
decrease in terminal half-life.
Nevirapine is mainly excreted in the urine as glucuronide
conjugates of the hydroxylated metabolites. |
| | Nevirapine Preparation Products And Raw materials |
|